

April 5, 2002
Bulletin #78
Table of contents:
Bulletin of the Month
Water Quality For Ostriches
By: Daryl Holle, Blue Mountain Feeds, Inc.
Our e-mail address
Subscribe – Unsubscribe information
Bulletin of the Month
Water Quality For Ostriches
Water is the most essential of all nutrients in the Ostrich diet. Approximately 60 to 85 percent (sometimes even higher) of the daily nutrition (water and feed) of farm livestock is represented by water. The fat-free adult body’s water content is relatively constant for many livestock species averaging 71 to 73 percent of body weight.
Water quality depends on proper construction, protection and maintenance of the entire water system, including the source.
Water quality directly affects water consumption. The first effect of water restriction, whether voluntary or involuntary, is reduced feed consumption with resulting lowered Ostrich productivity.
Palatability and Toxic Substances:
Low quality water may cause problems in two ways: Poor palatability may reduce consumption or toxic substances may be present. Some toxic substances do not reduce palatability and they are more harmful than those that do. Substances that may prove toxic in drinking water include pesticides, fluorine, molybdenum, nitrates, selenium and high concentrations of other specific elements.
Water may be contaminated with bacteria, viruses, protozoa or worm eggs, which can produce disease or other problems. In addition to toxic substances, water may contain other compounds that render it unpalatable, an example is alkali water containing high concentrations of Potassium, Sodium and Calcium Carbonates. Water high in saline is less palatable than non-mineral water. Saline salts are Sodium, Calcium, Magnesium and Potassium in the bicarbonate, chloride or sulfate form.
 |
A partial listing of some of the more common water contaminants are:
1. Total Dissolved Solids (Hardness):
This term includes all the minerals which have been dissolved as the water percolates downward through the soil and rock formations.
Most domestic animals can tolerate a total dissolved solid concentration in the range of 15,000 to 17,000 Parts Per Million (ppm). However, these concentrations will likely affect production. Salts in amounts of 5,000 ppm affect palatability for animals and, if consumed, will produce weight loss and diarrhea.
Sulfates, which are usually Magnesium sulfate (Epsom salt), sodium sulfate (Glaubers salt) or Calcium sulfate, cause a laxative effect in animals. Laxative effect is more pronounced in young than mature animals. The U.S. Public Health Services recommends that water containing more that 250 ppm of chlorides or sulfates and 500 ppm of dissolved solids are not to be used for human consumption. Sulfates over 500 ppm in water can cause scours in young animals.
Iron in concentrations as little as 0.3 ppm in water will cause brown staining on water fixtures and enclosures. It is not unusual to find up to 30 ppm of iron in some rural water wells. Ostrich are rather unique in the fact that they do not tolerate high intakes of Iron very well so care must be taken to eliminate any unnecessary Iron in the Ostrich diet. What level of Iron in the water that can be considered a Maximum level for Ostrich is difficult to determine as it totally depends on the Iron levels from all the other feed ingredients in the Ostrich daily diet. Ostrich also like to eat dirt on occasion and if the soil is high in Iron content, that is another source of Iron intake for the bird.
While it is highly unlikely that Ostrich can become toxic from too much Iron in water, feed and surrounding soil, it can be an excess factor that lowers the productivity of the birds. Too much Iron in the Ostrich diet, from whatever source, can cause an interference with the utilization of other important minerals and trace minerals in the Ostrich diet. Too much Iron in the Ostrich diet can specifically interfere with the utilization of Phosphorus, sometimes causing a deficiency of Phosphorus in the bird’s diet. Phosphorus is crucial to good growth (bone and muscle development) and also crucial to good fertility and reproduction along with many other things. In Ostrich, a deficiency of Phosphorus along with an excess of Iron has been conclusively determined to cause Ostrich meat to inherently have a “Liver” taste which is most objectionable to consumers.
Therefore, it is recommended by Blue Mountain Feeds to keep the Iron level in the water consumed by the birds to a level of 0.3 ppm. If your water is testing much higher than that recommended level, it is suggested that a water filtration system be installed that will remove the excess Iron and other contaminates.
Manganese often accompanies Iron and acts similarly except for the fact that high Manganese levels will usually leave a black stain on water fixtures and enclosures.
2. Nitrates:
Nitrates are widely dispersed in the environment and are particularly beneficial to plants. Health hazards can develop when significant amounts of nitrates (NO3) enter drinking water.
Nitrates (NO3) are reduced to a much more toxic product, nitrites (NO2), by bacteria in the intestinal trace and livestock waterers.
Nitrates and nitrites have an additional effect. When evaluating the levels to which animals are exposed, forages, bacterial action in waterers, as well as water amounts consumed, should be considered.
The 3 sources of nitrates and nitrites in drinking water are:
a. The percolating action of water passing through the soil.
b. Surface water or septic tank contamination of the well.
c. Bacterial breakdown of nitrates to nitrites in waterers.
The entire nitrate problem, its effect on production and reproduction, safe limits, complications with other nitrogen sources and desirable methods of removal or neutralization are all subjects of considerable controversy as it is a most complicated set of circumstances to properly ascertain.
Nitrates, if ingested in large quantities, can be converted by bacteria to nitrites, which are readily absorbed, and reduce the ability of the blood to carry oxygen (methemoglobin).
In some animal species, a nitrate problem may appear as a Vitamin A deficiency. This is one important reason why all Ostrich rations should most certainly have adequate amounts of Vitamin A, Iodine, Phosphorus, and Energy which helps fight off a nutritional imbalance from nitrates to a certain degree.
Nitrate levels in rural farm wells fluctuate widely. Usually wet spells in the spring season result in the highest levels of nitrates in rural farm water wells. Removal of nitrates from water is difficult and expensive as they are dissolved in water. Basic anion exchangers will remove nitrates.
The range of arbitrarily unsafe levels of nitrates in water probably lies somewhere between 50 ppm and 100 ppm. The apparent interference with normal nutrition, gestation, growth and health begins at about 100 ppm of nitrates (NO3). The U.S. Public Health Services sets 45 ppm of nitrates as the upper limit for a safe water supply for humans.
3. Microorganisms:
Coliform bacteria are nearly everywhere and may be plant, animal or soil origin. The term fecal coliform bacteria refers to normal organisms found in the gastrointestinal tract of livestock, humans, and birds. While these bacteria may not be harmful, their presence often indicates that other disease-causing bacteria and viruses may also be present.
Harmful microorganisms can readily enter a rural farm water well having improper protection from surface water contamination. It is possible that microorganisms can contaminate a water supply at the drinking point. Test result levels of 0 for coliform bacteria is satisfactory, 1 to 8 unsatisfactory, 9 or more is unsafe per M.P.N. (minimal probably number).
Prevention of surface water contamination, chlorination and filters help remedy microorganism problems.
4. pH:
Acid water corrodes pumps, pipes, tanks and fixtures. Acid water can be neutralized by adding soda ash or caustic soda to the water.
Alkalinity is found in most water supplies and comes from bicarbonates, carbonates and hydroxides giving a soda taste to the water. It usually promotes the growth of bacteria, and may cause scale to accumulate in water pipes and heaters.
5. Chlorides:
Alone, Chlorides are not harmful unless in large quantities over 200 ppm. Chlorides unite with Sodium, forming brine wells, and other salts, rendering some rural farm water wells useless. Levels of 50 ppm to 100 ppm are more palatable to livestock. Human water supplies that contain more than 5 ppm of Chlorides are objectionable.
6. Phosphates:
When phosphates are found in rural farm well water in large amounts, they usually indicate pollution, although they are generally harmless. They can serve as a source of nutrition for bacteria, and their presence should alert one to the probability of bacterial contamination.
7. Hydrogen Sulfide:
This is the “rotten egg” odor of water from deep water wells. As little as 1 ppm will produce an objectionable smell. The weak acid attacks Iron, forming a black, greasy deposit. The most economical method to remove hydrogen sulfide is oxidation by a chemical such as hypochloride, which will change the gas to elemental Sulfur that can be filtered out of the water.
8. Pesticides and Herbicides:
Pesticides or herbicides can enter ground water or surface water from run-off, drift, rainfall or accidental spills. Should a water supply become contaminated, notify the local government agency in charge of handling such misfortunes for instructions and guidelines.
9. Other Toxic Elements and Substances:
Whether from natural or human sources, water occasionally contains elements or substances in toxic amounts. Unfortunately, only limited information is available on experimentally determined toxic levels of various substances in water for livestock. So many conditions are involved in determining whether or not certain levels of a toxicant will cause harm that no single concentration can be accepted as dangerous in all situations. Toxic substances in water may be either a part of the suspended solids, be in solution or distributed between the two. Their availability in these phases may differ considerably during the digestion process within animals. Short-term intake of toxic substances may have no observable effect, while long-term consumption may result in serious harm. Different species of animals may react differently to a toxic substance, and the young and healthy may not respond in the same manner as mature or unthrifty animals.
It should be pointed out that water sources, especially those from shallow water wells, are subject to sudden changes in composition from natural or human causes.
A number of elements found in water seldom offer any problem to livestock because they do not occur at high levels in soluble form or because they are toxic only in excessive concentrations. Examples of these are Aluminum, Beryllium, Boron, Chromium, Cobalt, Copper, Iodine, Manganese, Molybdenum and Zinc. Also, these elements do not seem to accumulate in meat, milk or eggs to the extent that they would constitute a problem in livestock drinking water under any but the most unusual conditions.
On the other hand, elements such as Lead, Mercury and Cadmium must be considered actual or potential problems, because they occasionally are found in water at toxic concentrations or may accumulate in meat, milk or eggs at levels unfit for human consumption.
Water Testing
Contact your local Health Service, Agricultural Agent or Veterinarian for information on where water samples can be properly examined and what tests may be required. Get instructions on quantities, containers and handling for the required tests.
Following is a chart developed by the U.S. Public Health Service stating their feelings of proper and safe drinking water standards:
U.S. Public Health Service Drinking Water Standards
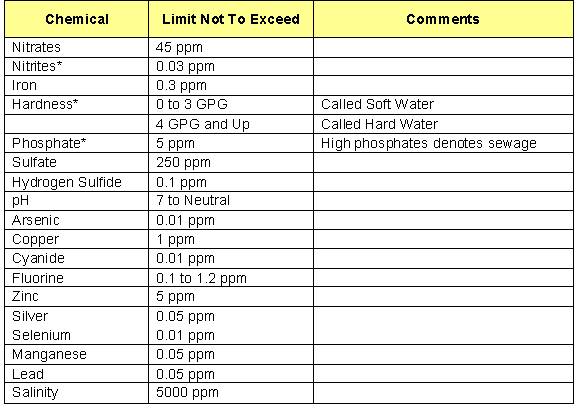
*Accepted by most laboratories
Conclusion
If your Ostrich water supply is from a rural farm water well, it is always wise to pay close attention to the water quality coming from your water well. Never assume that water quality is OKAY, always test and verify that there is nothing harmful to your birds. It is also wise to test the water during different seasons of the year as it can vary greatly depending on the circumstances occurring in your area.
Click to return to Contents Page
 |
Contact Information:
If you have any Bulletin Topics you would like us to consider for publication in a future E-Bulletin, e-mail your information to
. We are always interested in hearing your ideas about the value of this E-Bulletin, or whether you have any problems viewing this document.
If you would like to discontinue your subscription to this E-Bulletin, or if you have additional e-mail addresses to add to our subscriber list, please visit the following page on our website and use the automated form to “subscribe” or “unsubscribe”: www.blue-mountain.net/feed/bulletin.htm
Also check out our web site at www.blue-mountain.net for feature articles addressing Ostrich Nutrition, Feeding Management, and Ostrich Farm Management.
Click to return to Contents Page
 |
Blue Mountain Ostrich Nutrition E-Bulletin © April 2002
Return to Bulletin Menu